CES
-
 KAIST's Mathematician Reveals the Mechanism for Sustaining Biological Rhythms
Our bodies have a variety of biological clocks that follow rhythms or oscillations with periods ranging from seconds to days. For example, our hearts beat every second, and cells divide periodically. The circadian clock located in the hypothalamus generates twenty-four hour rhythms, timing our sleep and hormone release. How do these biological clocks or circuits generate and sustain the stable rhythms that are essential to life?
Jae Kyoung Kim, who is an assistant professor in the Department of Mathematical Sciences at KAIST, has predicted how these biological circuits generate rhythms and control their robustness, utilizing mathematical modeling based on differential equations and stochastic parameter sampling. Based on his prediction, using synthetic biology, a research team headed by Matthew Bennett of Rice University constructed a novel biological circuit that spans two genetically engineered strains of bacteria, one serves as an activator and the other as a repressor to regulate gene expression across multiple cell types, and found that the circuit generates surprisingly robust rhythms under various conditions.
The results of the research conducted in collaboration with KAIST (Korea Institute of Science and Technology), Rice University, and the University of Houston were published in Science (August 28, 2015 issue). The title of the paper is "Emergent Genetic Oscillations in a Synthetic Microbial Consortium" .
The top-down research approach, which focuses on identifying the components of biological circuits, limits our understanding of the mechanisms in which the circuits generate rhythms. Synthetic biology, a rapidly growing field at the interface of biosciences and engineering, however, uses a bottom-up approach.
Synthetic biologists can create complex circuits out of simpler components, and some of these new genetic circuits are capable of fluctuation to regulate gene production. In the same way that electrical engineers understand how an electrical circuit works as they construct batteries, resistors, and wires, synthetic biologists can understand better about biological circuits if they put them together using genes and proteins. However, due to the complexity of biological systems, both experiments and mathematical modeling need to be applied hand in hand to design these biological circuits and understand their function.
In this research, an interdisciplinary approach proved that a synthetic intercellular singling circuit generates robust rhythms to create a cooperative microbial system. Specifically, Kim's mathematical analysis suggested, and experiments confirmed, that the presence of negative feedback loops in addition to a core transcriptional negative feedback loop can explain the robustness of rhythms in this system. This result provides important clues about the fundamental mechanism of robust rhythm generation in biological systems.
Furthermore, rather than constructing the entire circuit inside a single bacterial strain, the circuit was split among two strains of Escherichia coli bacterium. When the strains were grown together, the bacteria exchanged information, completing the circuit. Thus, this research also shows how, by regulating individual cells within the system, complex biological systems can be controlled, which in turn influences each other (e.g., the gut microbiome in humans).
###
Ye Chen, a graduate student in Bennett's laboratory at Rice University, and Jae Kyoung Kim, an assistant professor at KAIST and a former postdoctoral fellow at Ohio State University, are the lead authors of the paper. The co-authors are Rice graduate student Andrew Hirning and Krešimir Josic?, a professor of mathematics at the University of Houston. Bennett is the Assistant Professor of the Biochemistry and Cell Biology Department at Rice University.
About the researcher: While Jae Kyoung Kim is a mathematician, he has also solved various biological puzzles in collaboration with various experimental laboratories of Matthew Bennett at Rice University, David Virshup at Duke and the National University of Singapore, Carla Finkielstein at Virginia Polytechnic Institute and State University, Choo-Gon Lee at the Florida State University, Seung-Hee Yoo at the Medical School of the University of Texas, Toru Takumi at RIKEN Brain Science Institute, and Travis Wager at Pfizer Inc. He has used non-linear dynamics and stochastic analysis to understand the function of biochemical networks in biological systems. In particular, he is interested in mechanisms generating and regulating biological rhythms.
Picture 1: This schematic image is the design of a biological circuit between two strains of bacteria and the part of differential equations used to understand the function of the biological circuit.
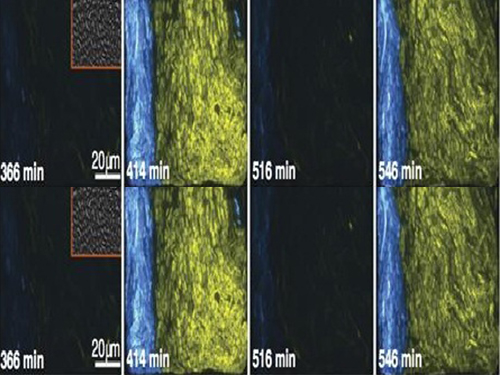
Picture 2: The core transcriptional negative feedback loop and additional negative feedback loop in the biological circuit (picture 1) generate robust rhythms. The snapshots correspond the red dots in the time series graph.
2015.08.31 View 9716
KAIST's Mathematician Reveals the Mechanism for Sustaining Biological Rhythms
Our bodies have a variety of biological clocks that follow rhythms or oscillations with periods ranging from seconds to days. For example, our hearts beat every second, and cells divide periodically. The circadian clock located in the hypothalamus generates twenty-four hour rhythms, timing our sleep and hormone release. How do these biological clocks or circuits generate and sustain the stable rhythms that are essential to life?
Jae Kyoung Kim, who is an assistant professor in the Department of Mathematical Sciences at KAIST, has predicted how these biological circuits generate rhythms and control their robustness, utilizing mathematical modeling based on differential equations and stochastic parameter sampling. Based on his prediction, using synthetic biology, a research team headed by Matthew Bennett of Rice University constructed a novel biological circuit that spans two genetically engineered strains of bacteria, one serves as an activator and the other as a repressor to regulate gene expression across multiple cell types, and found that the circuit generates surprisingly robust rhythms under various conditions.
The results of the research conducted in collaboration with KAIST (Korea Institute of Science and Technology), Rice University, and the University of Houston were published in Science (August 28, 2015 issue). The title of the paper is "Emergent Genetic Oscillations in a Synthetic Microbial Consortium" .
The top-down research approach, which focuses on identifying the components of biological circuits, limits our understanding of the mechanisms in which the circuits generate rhythms. Synthetic biology, a rapidly growing field at the interface of biosciences and engineering, however, uses a bottom-up approach.
Synthetic biologists can create complex circuits out of simpler components, and some of these new genetic circuits are capable of fluctuation to regulate gene production. In the same way that electrical engineers understand how an electrical circuit works as they construct batteries, resistors, and wires, synthetic biologists can understand better about biological circuits if they put them together using genes and proteins. However, due to the complexity of biological systems, both experiments and mathematical modeling need to be applied hand in hand to design these biological circuits and understand their function.
In this research, an interdisciplinary approach proved that a synthetic intercellular singling circuit generates robust rhythms to create a cooperative microbial system. Specifically, Kim's mathematical analysis suggested, and experiments confirmed, that the presence of negative feedback loops in addition to a core transcriptional negative feedback loop can explain the robustness of rhythms in this system. This result provides important clues about the fundamental mechanism of robust rhythm generation in biological systems.
Furthermore, rather than constructing the entire circuit inside a single bacterial strain, the circuit was split among two strains of Escherichia coli bacterium. When the strains were grown together, the bacteria exchanged information, completing the circuit. Thus, this research also shows how, by regulating individual cells within the system, complex biological systems can be controlled, which in turn influences each other (e.g., the gut microbiome in humans).
###
Ye Chen, a graduate student in Bennett's laboratory at Rice University, and Jae Kyoung Kim, an assistant professor at KAIST and a former postdoctoral fellow at Ohio State University, are the lead authors of the paper. The co-authors are Rice graduate student Andrew Hirning and Krešimir Josic?, a professor of mathematics at the University of Houston. Bennett is the Assistant Professor of the Biochemistry and Cell Biology Department at Rice University.
About the researcher: While Jae Kyoung Kim is a mathematician, he has also solved various biological puzzles in collaboration with various experimental laboratories of Matthew Bennett at Rice University, David Virshup at Duke and the National University of Singapore, Carla Finkielstein at Virginia Polytechnic Institute and State University, Choo-Gon Lee at the Florida State University, Seung-Hee Yoo at the Medical School of the University of Texas, Toru Takumi at RIKEN Brain Science Institute, and Travis Wager at Pfizer Inc. He has used non-linear dynamics and stochastic analysis to understand the function of biochemical networks in biological systems. In particular, he is interested in mechanisms generating and regulating biological rhythms.
Picture 1: This schematic image is the design of a biological circuit between two strains of bacteria and the part of differential equations used to understand the function of the biological circuit.
Picture 2: The core transcriptional negative feedback loop and additional negative feedback loop in the biological circuit (picture 1) generate robust rhythms. The snapshots correspond the red dots in the time series graph.
2015.08.31 View 9716 -
 Dr. Se-Jung Kim Receives the Grand Prize at the International Photo and Image Contest on Light
Dr. Se-Jung Kim of the Physics Department at KAIST received the Grand Prize at the 2015 Photo and Image Contest of the International Year of Light and Light-based Technologies.
The United Nations has designated the year 2015 as the International Year of Light and Light-based Technologies.
The Optical Society of Korea celebrated the UN’s designation by hosting an international photo and image contest on the theme of light and optics related technology.
Dr. Kim presented a photo of images taken from a liquid crystal, which was entitled “A Micro Pinwheel.” She took pictures of liquid crystal images with a polarizing microscope and then colored the pictures. The liquid crystal has self-assembled circle domain structures, and each domain can form vortex optics. Her adviser for the project is Professor Yong-Hoon Cho of the Physics Department.
Her work was exhibited during the annual conference of the Optical Society of Korea, which was held on July 13-15, 2015 at Gyeong-Ju Hwabaek International Convention Center. It will also be exhibited at the National Science Museum in Gwacheon and the Kim Dae-Jung Convention Center in Gwangju.
Picture: A Micro Pinwheel
2015.07.31 View 13171
Dr. Se-Jung Kim Receives the Grand Prize at the International Photo and Image Contest on Light
Dr. Se-Jung Kim of the Physics Department at KAIST received the Grand Prize at the 2015 Photo and Image Contest of the International Year of Light and Light-based Technologies.
The United Nations has designated the year 2015 as the International Year of Light and Light-based Technologies.
The Optical Society of Korea celebrated the UN’s designation by hosting an international photo and image contest on the theme of light and optics related technology.
Dr. Kim presented a photo of images taken from a liquid crystal, which was entitled “A Micro Pinwheel.” She took pictures of liquid crystal images with a polarizing microscope and then colored the pictures. The liquid crystal has self-assembled circle domain structures, and each domain can form vortex optics. Her adviser for the project is Professor Yong-Hoon Cho of the Physics Department.
Her work was exhibited during the annual conference of the Optical Society of Korea, which was held on July 13-15, 2015 at Gyeong-Ju Hwabaek International Convention Center. It will also be exhibited at the National Science Museum in Gwacheon and the Kim Dae-Jung Convention Center in Gwangju.
Picture: A Micro Pinwheel
2015.07.31 View 13171 -
 Professor Sangyong Jon Appointed Fellow of AIMBE
Professor Sangyong Jon of the Department of Biological Sciences at KAIST has been appointed a member of the American Institute for Medical and Biological Engineering (AIMBE) fellowship.
Established in 1991, AIMBE is a non-profit organization based in Washington, D.C., representing 50,000 individuals and the top 2% of medical and biological engineers. AIMBE provides policy advice and advocacy for medical and biological engineering for the benefit of humanity. It has had about 1,500 fellows over the past 25 years. Among the members, only 110 are non-American nationalities.
Following the appointment of Dr. Hae-Bang Lee, the former senior researcher at the Korean Research Institute of Chemical Technology, and Distinguished Professor Sang Yup Lee of the Department of Chemical and Biomolecular Engineering at KAIST, Professor Jon is the third Korean to become an AIMBE fellow. He had an induction ceremony for the appointment of his fellowship at the AIMBE’s Annual Event held on March 15-17, 2015 in Washington, D.C.
An authority on nanomedicine, Professor Jon has developed many original technologies including multi-functional Theranostics nano particles for the diagnosis and treatment of diseases. He received the Most Cited Paper Award from Theranostics, an academic journal specialized in nanomedicine, last February.
Additionally, Professor Jon is a leading researcher in the field of translational medicine, using a multi-disciplinary, highly collaborative, “Bench to Bedside” approach for disease treatment and prevention. He created a biotechnology venture company and transferred research developments to the industry in Korea.
2015.03.12 View 15040
Professor Sangyong Jon Appointed Fellow of AIMBE
Professor Sangyong Jon of the Department of Biological Sciences at KAIST has been appointed a member of the American Institute for Medical and Biological Engineering (AIMBE) fellowship.
Established in 1991, AIMBE is a non-profit organization based in Washington, D.C., representing 50,000 individuals and the top 2% of medical and biological engineers. AIMBE provides policy advice and advocacy for medical and biological engineering for the benefit of humanity. It has had about 1,500 fellows over the past 25 years. Among the members, only 110 are non-American nationalities.
Following the appointment of Dr. Hae-Bang Lee, the former senior researcher at the Korean Research Institute of Chemical Technology, and Distinguished Professor Sang Yup Lee of the Department of Chemical and Biomolecular Engineering at KAIST, Professor Jon is the third Korean to become an AIMBE fellow. He had an induction ceremony for the appointment of his fellowship at the AIMBE’s Annual Event held on March 15-17, 2015 in Washington, D.C.
An authority on nanomedicine, Professor Jon has developed many original technologies including multi-functional Theranostics nano particles for the diagnosis and treatment of diseases. He received the Most Cited Paper Award from Theranostics, an academic journal specialized in nanomedicine, last February.
Additionally, Professor Jon is a leading researcher in the field of translational medicine, using a multi-disciplinary, highly collaborative, “Bench to Bedside” approach for disease treatment and prevention. He created a biotechnology venture company and transferred research developments to the industry in Korea.
2015.03.12 View 15040 -
 The Number of KAIST Doctoral Graduates to Reach Over Ten Thousands
The ten-thousandth doctoral graduate received her degree in the commencement ceremony on February 13, 2015.
KAIST has contributed to the development of science, technology, and industry in Korea by fostering talents in advanced science and engineering.
Since its establishment forty-four years ago, more than ten-thousand KAIST alumni have received their doctorates. This year’s graduation ceremony took place on February 13, 2015, at the Sports Complex on campus, awarding the ten-thousandth doctoral degree.
Dr. Sun-Mi Cho of the Department of Biological Sciences received the ten-thousandth doctoral degree. A graduate of Jeon-Nam Science High School, Dr. Cho also received her Bachelor of Science degree from KAIST.
Dr. Cho wrote a dissertation entitled “GABA from reactive astrocytes impairs learning and memory in Alzheimer disease.” Her dissertation adviser was Professor Daesoo Kim of the Department of Biological Sciences.
Dr. Cho, who will be a post-doctorate researcher at the Biological Sciences Department, said, “It was my childhood dream to receive a doctorate from KAIST. I cannot believe that I’m the ten-thousandth doctoral graduate, for which I’m very grateful.” She continued, “I hope to become a neuroscientist where I can be of help to the sick.”
In 1978, KAIST only had two doctoral graduates, but since 1987 there have been more than one hundred graduates each year, two hundred since 1994, and four hundred since 2000. In 2015, 522 doctoral students graduated.
One of the first doctoral graduates, Dong-Yol Yang (class of 1978 in the Mechanical Engineering Department), became a professor at the same department of KAIST. Professor Yang expressed his thoughts on the news, “There was a trend in Korea to go overseas for Ph.D. degrees in the early 1970s, but it changed when KAIST began to select candidates for Master’s degrees in 1973 and Doctoral degrees in 1975. Talented Korean students came to KAIST laboratories, and its graduates were known for their knowledge and skills. Now, we see that the talent is coming from overseas.”
At the 2015 Commencement, KAIST conferred 522 Doctoral, 1,241 Master’s, and 915 Bachelor of Science (B.S.) degrees.
Since its inception in 1971, KAIST has granted 10,403 doctor's, 26,402 master's, and 51,412 bachelor's degrees.
In the picture below, Professor Dong-Yol Yang (left) seats next to Dr. Sun-Mi Cho (right), the recipient of 10,000th doctoral degree.
2015.02.16 View 11228
The Number of KAIST Doctoral Graduates to Reach Over Ten Thousands
The ten-thousandth doctoral graduate received her degree in the commencement ceremony on February 13, 2015.
KAIST has contributed to the development of science, technology, and industry in Korea by fostering talents in advanced science and engineering.
Since its establishment forty-four years ago, more than ten-thousand KAIST alumni have received their doctorates. This year’s graduation ceremony took place on February 13, 2015, at the Sports Complex on campus, awarding the ten-thousandth doctoral degree.
Dr. Sun-Mi Cho of the Department of Biological Sciences received the ten-thousandth doctoral degree. A graduate of Jeon-Nam Science High School, Dr. Cho also received her Bachelor of Science degree from KAIST.
Dr. Cho wrote a dissertation entitled “GABA from reactive astrocytes impairs learning and memory in Alzheimer disease.” Her dissertation adviser was Professor Daesoo Kim of the Department of Biological Sciences.
Dr. Cho, who will be a post-doctorate researcher at the Biological Sciences Department, said, “It was my childhood dream to receive a doctorate from KAIST. I cannot believe that I’m the ten-thousandth doctoral graduate, for which I’m very grateful.” She continued, “I hope to become a neuroscientist where I can be of help to the sick.”
In 1978, KAIST only had two doctoral graduates, but since 1987 there have been more than one hundred graduates each year, two hundred since 1994, and four hundred since 2000. In 2015, 522 doctoral students graduated.
One of the first doctoral graduates, Dong-Yol Yang (class of 1978 in the Mechanical Engineering Department), became a professor at the same department of KAIST. Professor Yang expressed his thoughts on the news, “There was a trend in Korea to go overseas for Ph.D. degrees in the early 1970s, but it changed when KAIST began to select candidates for Master’s degrees in 1973 and Doctoral degrees in 1975. Talented Korean students came to KAIST laboratories, and its graduates were known for their knowledge and skills. Now, we see that the talent is coming from overseas.”
At the 2015 Commencement, KAIST conferred 522 Doctoral, 1,241 Master’s, and 915 Bachelor of Science (B.S.) degrees.
Since its inception in 1971, KAIST has granted 10,403 doctor's, 26,402 master's, and 51,412 bachelor's degrees.
In the picture below, Professor Dong-Yol Yang (left) seats next to Dr. Sun-Mi Cho (right), the recipient of 10,000th doctoral degree.
2015.02.16 View 11228 -
 Professor Eunjoon Kim Is KAIST's Person of the Year 2014
KAIST announced that it has named Chair Professor Eunjoon Kim of the Department of Biological Sciences as its “Person of the Year 2014.” The award ceremony took place at the auditorium on campus on January 5, 2014.
Established in 2001, the award has been presented to a KAIST faculty member who has made great achievements in research and education, thereby contributing to the advancement of KAIST.
Professor Kim was the first to identify the mechanism of synapse formation between neurons during his post-doctoral program at Harvard Medical School in 1995. The research was published in Nature.
In 2011, Professor Kim discovered that the lack of protein GIT1, a neuronal synapse in the brain, caused ADHD (Attention Deficit Hyperactivity Disorder). He is widely recognized for his work concerning synapse proteins and brain disease related research that set the foundation for future medical developments.
In his award speech, Professor Kim said, “Whenever a research finding concerning a new drug therapy or research is published, I receive many inquiries from the parents of children with ADHD or autism. As a scientist, I would like to focus my research ultimately to help those in pain, rather than just pursuing research excellence or reputation.”
2015.01.06 View 11961
Professor Eunjoon Kim Is KAIST's Person of the Year 2014
KAIST announced that it has named Chair Professor Eunjoon Kim of the Department of Biological Sciences as its “Person of the Year 2014.” The award ceremony took place at the auditorium on campus on January 5, 2014.
Established in 2001, the award has been presented to a KAIST faculty member who has made great achievements in research and education, thereby contributing to the advancement of KAIST.
Professor Kim was the first to identify the mechanism of synapse formation between neurons during his post-doctoral program at Harvard Medical School in 1995. The research was published in Nature.
In 2011, Professor Kim discovered that the lack of protein GIT1, a neuronal synapse in the brain, caused ADHD (Attention Deficit Hyperactivity Disorder). He is widely recognized for his work concerning synapse proteins and brain disease related research that set the foundation for future medical developments.
In his award speech, Professor Kim said, “Whenever a research finding concerning a new drug therapy or research is published, I receive many inquiries from the parents of children with ADHD or autism. As a scientist, I would like to focus my research ultimately to help those in pain, rather than just pursuing research excellence or reputation.”
2015.01.06 View 11961 -
 Professor Mikyoung Lim Receives the MediaV Young Researcher Award
Professor Mikyoung Lim of the Department of Mathematical Sciences at KAIST received the MediaV Young Researchers Award at the International Conference on Inverse Problems and Related Topics that took place at the National Taiwan University, Taiwan, on December 15-19, 2014.
The Conference established the MediaV Young Researcher Award in 2010 to recognize distinguished scholars who are age 40 or younger and have made important contributions to the field of inverse problems. This year, two recipients were chosen for the award.
Professor Lim has focused her research on the incremental reading of incomprehensible materials’ imaging and the effect of invisibility cloaking.
The other awardee was Kui Ren, a professor at the University of Texas at Austin.
2014.12.27 View 13589
Professor Mikyoung Lim Receives the MediaV Young Researcher Award
Professor Mikyoung Lim of the Department of Mathematical Sciences at KAIST received the MediaV Young Researchers Award at the International Conference on Inverse Problems and Related Topics that took place at the National Taiwan University, Taiwan, on December 15-19, 2014.
The Conference established the MediaV Young Researcher Award in 2010 to recognize distinguished scholars who are age 40 or younger and have made important contributions to the field of inverse problems. This year, two recipients were chosen for the award.
Professor Lim has focused her research on the incremental reading of incomprehensible materials’ imaging and the effect of invisibility cloaking.
The other awardee was Kui Ren, a professor at the University of Texas at Austin.
2014.12.27 View 13589 -
 Nanoparticle Cluster Manufacturing Technique Using DNA Binding Protein Developed
Professor Hak-Sung Kim of the Department of Biological Sciences at KAIST and Yiseul Ryu, a doctoral candidate, used the Zinc Finger protein that specifically binds to target DNA sequence to develop a new manufacturing technique for size-controllable magnetic Nanoparticle Clusters (NPCs). Their research results were published in Angewandte Chemie International Edition online on 25 November 2014.
NPCs are structures consisting of magnetic nanoparticles, gold nanoparticles, and quantum dots, each of which are smaller than 100 nm (10-9m). NPCs have a distinctive property of collectivity not seen in single nanoparticles.
Specifically NPCS differ in physical and optical properties such as Plasmon coupling absorbance, energy transfers between particles, electron transfers, and conductivity. Therefore, NPCs can be employed in biological and medical research as well as the development of nanoelectric and nanoplasmon devices.
To make use of these novel properties, the size and the composition of the cluster must be exquisitely controlled. However, previous techniques relied on chemical binding which required complex steps, making it difficult to control the size and composition of NPCs.
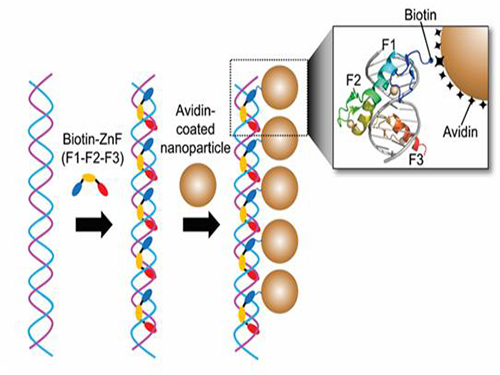
Professor Kim’s team used Zinc Finger, a DNA binding protein, to develop a NPCs manufacturing technique to create clusters of the desired size easily. The Zinc Finger protein contains a zinc ion and specifically recognizes DNA sequence upon binding, which allows the exquisite control of the size and the cluster composition. The technique is also bio-friendly.
Professor Kim’s team created linear structure of different sizes of NPCs using Zinc Finger proteins and three DNA sequences of different lengths. The NPCs they produced confirmed their ability to control the size and structure of the cluster by using different DNA lengths.
The NPCs showed tripled T2 relaxation rates compared to the existing MRI contrast media (Feridex) and effectively transported to targeted cells. The research findings show the potential use of NPCs in biological and medical fields such as MRI contrast media, fluorescence imaging, and drug transport.
The research used the specific binding property of protein and DNA to develop a new method to create an inorganic nanoparticle’s supramolecular assembly. The technique can be used and applied extensively in other nanoparticles for future research in diagnosis, imaging, and drug and gene delivery.
Figure 1. A Mimetic Diagram of NPCs Manufacturing Technique Using DNA Binding Protein Zinc Finger
Figure 2. Transmission Electron Microscopy Images showing different sizes of NPCs depending on the length of the DNA
2014.12.04 View 15358
Nanoparticle Cluster Manufacturing Technique Using DNA Binding Protein Developed
Professor Hak-Sung Kim of the Department of Biological Sciences at KAIST and Yiseul Ryu, a doctoral candidate, used the Zinc Finger protein that specifically binds to target DNA sequence to develop a new manufacturing technique for size-controllable magnetic Nanoparticle Clusters (NPCs). Their research results were published in Angewandte Chemie International Edition online on 25 November 2014.
NPCs are structures consisting of magnetic nanoparticles, gold nanoparticles, and quantum dots, each of which are smaller than 100 nm (10-9m). NPCs have a distinctive property of collectivity not seen in single nanoparticles.
Specifically NPCS differ in physical and optical properties such as Plasmon coupling absorbance, energy transfers between particles, electron transfers, and conductivity. Therefore, NPCs can be employed in biological and medical research as well as the development of nanoelectric and nanoplasmon devices.
To make use of these novel properties, the size and the composition of the cluster must be exquisitely controlled. However, previous techniques relied on chemical binding which required complex steps, making it difficult to control the size and composition of NPCs.
Professor Kim’s team used Zinc Finger, a DNA binding protein, to develop a NPCs manufacturing technique to create clusters of the desired size easily. The Zinc Finger protein contains a zinc ion and specifically recognizes DNA sequence upon binding, which allows the exquisite control of the size and the cluster composition. The technique is also bio-friendly.
Professor Kim’s team created linear structure of different sizes of NPCs using Zinc Finger proteins and three DNA sequences of different lengths. The NPCs they produced confirmed their ability to control the size and structure of the cluster by using different DNA lengths.
The NPCs showed tripled T2 relaxation rates compared to the existing MRI contrast media (Feridex) and effectively transported to targeted cells. The research findings show the potential use of NPCs in biological and medical fields such as MRI contrast media, fluorescence imaging, and drug transport.
The research used the specific binding property of protein and DNA to develop a new method to create an inorganic nanoparticle’s supramolecular assembly. The technique can be used and applied extensively in other nanoparticles for future research in diagnosis, imaging, and drug and gene delivery.
Figure 1. A Mimetic Diagram of NPCs Manufacturing Technique Using DNA Binding Protein Zinc Finger
Figure 2. Transmission Electron Microscopy Images showing different sizes of NPCs depending on the length of the DNA
2014.12.04 View 15358 -
 Thomson Reuters Nominates Distinguished Professor Ryong Ryoo for Its 2014 Nobel Citation Laureates in Chemistry
The Intellectual Property & Science business of Thomson Reuters announced on September 25th its “2014 Citation Laureates,” a list of candidates considered likely to win the Nobel Prize in the fields of physics, chemistry, physiology or medicine, and economics. The annual Thomson Reuters Citation Laureates will be recognized in perpetuity as contenders for a Nobel Prize.
Distinguished Professor Ryong Ryoo of the Department of Chemistry, KAIST, has been nominated for the 2014 Thomson Reuters Citation Laureates in Chemistry. He is the first Korean scientist who has made the list. In addition to Professor Ryoo, seven other scientists were selected as possible contenders for the 2014 Nobel Prize in Chemistry, or in the future.
Professor Ryoo was named alongside Charles T. Kresge, Chief Technology Officer of Saudi Aramco, Dhahran, and Galen D. Stucky, Professor of the Department Chemistry and Biochemistry at the University of California, Santa Barbara, for their research on the design of functional mesoporous materials (http://sciencewatch.com/nobel/2014-predictions/chemistry-laureates). Mesoporous materials have high surface areas with narrow pore-sized distribution and tunable pores diameters, offering promising properties and applications in various areas including adsorption, separation, sensing, and catalysis.
Professor Ryoo has focused his research interest in the synthesis of new functional nanoporous materials such as hierarchical zeolites, mesoporous silicas, carbons, and organic-inorganic composite materials that can be used for advanced applications in the production of alternative energy sources and in green chemical processes.
According to the press release by the Thomson Reuters, the list of the 2014 Nobel predictions includes 27 researchers representing 27 distinct academic and research organizations across nine different countries.
The annual Thomson Reuters Citation Laureates study is based on the analysis of proprietary data from the research and citation database, identifying the most influential researchers in the categories of chemistry, physics, physiology or medicine, and economics. Since its inception in 2002, the study has accurately forecasted 35 Nobel Prize winners. For the full text of the press release, please go to: http://thomsonreuters.com/press-releases/092014/2014-nobel-laureates-predictions.
2014.09.29 View 13078
Thomson Reuters Nominates Distinguished Professor Ryong Ryoo for Its 2014 Nobel Citation Laureates in Chemistry
The Intellectual Property & Science business of Thomson Reuters announced on September 25th its “2014 Citation Laureates,” a list of candidates considered likely to win the Nobel Prize in the fields of physics, chemistry, physiology or medicine, and economics. The annual Thomson Reuters Citation Laureates will be recognized in perpetuity as contenders for a Nobel Prize.
Distinguished Professor Ryong Ryoo of the Department of Chemistry, KAIST, has been nominated for the 2014 Thomson Reuters Citation Laureates in Chemistry. He is the first Korean scientist who has made the list. In addition to Professor Ryoo, seven other scientists were selected as possible contenders for the 2014 Nobel Prize in Chemistry, or in the future.
Professor Ryoo was named alongside Charles T. Kresge, Chief Technology Officer of Saudi Aramco, Dhahran, and Galen D. Stucky, Professor of the Department Chemistry and Biochemistry at the University of California, Santa Barbara, for their research on the design of functional mesoporous materials (http://sciencewatch.com/nobel/2014-predictions/chemistry-laureates). Mesoporous materials have high surface areas with narrow pore-sized distribution and tunable pores diameters, offering promising properties and applications in various areas including adsorption, separation, sensing, and catalysis.
Professor Ryoo has focused his research interest in the synthesis of new functional nanoporous materials such as hierarchical zeolites, mesoporous silicas, carbons, and organic-inorganic composite materials that can be used for advanced applications in the production of alternative energy sources and in green chemical processes.
According to the press release by the Thomson Reuters, the list of the 2014 Nobel predictions includes 27 researchers representing 27 distinct academic and research organizations across nine different countries.
The annual Thomson Reuters Citation Laureates study is based on the analysis of proprietary data from the research and citation database, identifying the most influential researchers in the categories of chemistry, physics, physiology or medicine, and economics. Since its inception in 2002, the study has accurately forecasted 35 Nobel Prize winners. For the full text of the press release, please go to: http://thomsonreuters.com/press-releases/092014/2014-nobel-laureates-predictions.
2014.09.29 View 13078 -
 News Article on the Development of Synthesis Process for Graphene Quantum Dots
Before It's News, an international online news agency, highlighted the recent research conducted by KAIST professors (Seokwoo Jeon of the Department of Materials Science and Engineering, Yong-Hoon Cho of the Department of Physics, and Seunghyup Yoo of the Department of Electrical Engineering) on the development of synthesis process for graphene quantum dots, nanometer-sized round semiconductor nanoparticles that are very efficient at emitting photons. If commercialized, this synthetic technology will lead the way to the development of paper-thin displays in the future.
For the article, please go to the link below:
Before It’s News, September 3, 2014“Graphene quantum dots prove highly efficient in emitting light”
http://beforeitsnews.com/science-and-technology/2014/09/graphene-quantum-dots-prove-highly-efficient-in-emitting-light-2718190.html
2014.09.07 View 16389
News Article on the Development of Synthesis Process for Graphene Quantum Dots
Before It's News, an international online news agency, highlighted the recent research conducted by KAIST professors (Seokwoo Jeon of the Department of Materials Science and Engineering, Yong-Hoon Cho of the Department of Physics, and Seunghyup Yoo of the Department of Electrical Engineering) on the development of synthesis process for graphene quantum dots, nanometer-sized round semiconductor nanoparticles that are very efficient at emitting photons. If commercialized, this synthetic technology will lead the way to the development of paper-thin displays in the future.
For the article, please go to the link below:
Before It’s News, September 3, 2014“Graphene quantum dots prove highly efficient in emitting light”
http://beforeitsnews.com/science-and-technology/2014/09/graphene-quantum-dots-prove-highly-efficient-in-emitting-light-2718190.html
2014.09.07 View 16389 -
 Discovery of New Therapeutic Targets for Alzheimer's Disease
A Korean research team headed by Professor Dae-Soo Kim of Biological Sciences at KAIST and Dr. Chang-Jun Lee from the Korea Institute of Science and Technology (KIST) successfully identified that reactive astrocytes, commonly observed in brains affected by Alzheimer’s disease, produce abnormal amounts of inhibitory neurotransmitter gamma-Aminobutyric acid (GABA) in reaction to the enzyme Monoamine oxidase B (Mao-B) and release GABA through the Bestrophin-1 channel to suppress the normal signal transmission of brain nerve cells.
By suppressing the GABA production or release from reactive astrocytes, the research team was able to restore the model mice's memory and learning impairment caused by Alzheimer’s disease. This discovery will allow the development of new drugs to treat Alzheimer’s and other related diseases.
The research result was published in the June 29, 2014 edition of Nature Medicine (Title: GABA from Reactive Astrocytes Impairs Memory in Mouse Models of Alzheimer’s Disease).
For details, please read the article below:
Technology News, July 10, 2014
"Discovery of New Drug Targets for Memory Impairment in Alzheimer’s Disease"
http://technews.tmcnet.com/news/2014/07/10/7917811.htm
2014.07.16 View 10945
Discovery of New Therapeutic Targets for Alzheimer's Disease
A Korean research team headed by Professor Dae-Soo Kim of Biological Sciences at KAIST and Dr. Chang-Jun Lee from the Korea Institute of Science and Technology (KIST) successfully identified that reactive astrocytes, commonly observed in brains affected by Alzheimer’s disease, produce abnormal amounts of inhibitory neurotransmitter gamma-Aminobutyric acid (GABA) in reaction to the enzyme Monoamine oxidase B (Mao-B) and release GABA through the Bestrophin-1 channel to suppress the normal signal transmission of brain nerve cells.
By suppressing the GABA production or release from reactive astrocytes, the research team was able to restore the model mice's memory and learning impairment caused by Alzheimer’s disease. This discovery will allow the development of new drugs to treat Alzheimer’s and other related diseases.
The research result was published in the June 29, 2014 edition of Nature Medicine (Title: GABA from Reactive Astrocytes Impairs Memory in Mouse Models of Alzheimer’s Disease).
For details, please read the article below:
Technology News, July 10, 2014
"Discovery of New Drug Targets for Memory Impairment in Alzheimer’s Disease"
http://technews.tmcnet.com/news/2014/07/10/7917811.htm
2014.07.16 View 10945 -
 Artificial Antibody-based Therapeutic Candidate for Lung Cancer Developed
Professor Hak-Sung Kim of Biological Sciences at KAIST publishes a cover article on artificial antibody in "Molecular Therapy".
Repebody-based lung cancer therapeutic drug candidate developed Repebody-based protein demonstrates the possibility of the development of a new drug
KAIST Biological Sciences Department’s Professor Hak-Sung Kim, in collaboration with Professor Eun-Kyung Cho from the College of Medicine at Chungnam National University, has successfully developed an artificial antibody-based, or repebody, cancer therapeutic candidate. These research results were published as a cover paper of the July edition of Molecular Therapy.
The repebody developed by Professor Kim and his team strongly binds to interleukin-6, a cancer-causing factor. It has also been confirmed that the repebody can significantly inhibit the proliferation of cancer cells in non-small-cell lung cancer animal model.
Numerous multinational pharmaceutical and biotechnology companies have invested astronomical amounts of money in research for the development of protein therapeutics with low side effects and high efficacy. More than 20 kinds of such therapeutics are currently under clinical trials, and over 100 drugs are under clinical demonstration. Among these, the majority is antibody-based therapeutics, and most of the investments are heavily concentrated in this field. However, antibody production cost is very high because it has large molecular weights and complex structural properties, and this makes it difficult to engineer. Consequently, the development costs a great deal of time and money.
In order to overcome the existing limitations of antibody-based therapeutics, Professor Kim and his team have developed a new artificial antibody, or repebody, which was published in Proceedings of the National Academy of Sciences (PNAS) in 2012. Based on this research, they have succeeded in developing a therapeutic candidate for treating non-small-cell lung cancer with a specifically strong cohesion to the cancer-causing factor, interleukin-6.
Interleukin-6 is a crucial substance within the body that is involved in immune and inflammatory-related signals. When abnormally expressed, it activates various carcinogenic pathways and promotes tumor growth and metastasis. Because of its importance, multinational pharmaceutical companies are heavily investing in developing therapeutics that can inhibit the signaling of interleukin-6.
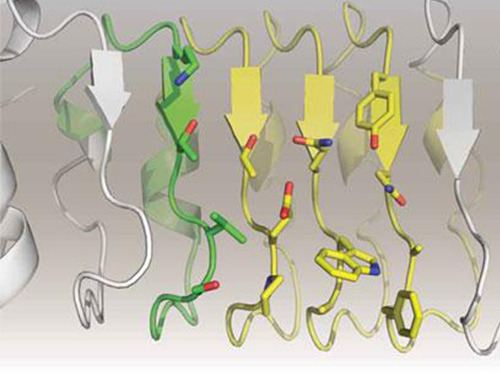
In this study, Professor Kim and his team observed that a repebody consists of repeated modules, and they conceived a module-based affinity amplification technology that can effectively increase the binding affinity with the disease target. The developed therapeutic candidate has been confirmed in cell and animal experiments to show low immunogenicity, as well as to strongly inhibit the proliferation of non-small-cell lung cancer.
Furthermore, by investigating the complex structure of the repebody with interleukin-6, Professor Kim has identified its mechanism, which demonstrated the potential for therapeutic development. The researchers are currently carrying out pre-clinical trials for acquiring permission to perform clinical trials on animals with non-small-cell lung cancer. The repebody can be developed into a new protein drug after demonstrating its safety and efficacy.
Professor Hak-Sung Kim and his team have confirmed that the repebody can be utilized as a new protein drug, and this will be a significant contribution to Korea’s protein drugs and biotechnology industry development.
The research was supported by the Future Pioneer Industry project and sponsored by the Ministry of Science, ICT and Future Planning.
Figure 1. Professor Kim’s article published as the cover article of July edition of Molecular Therapy
Figure 2. Clinical proof of the repebody’s inhibition of cancer growth using animal models
2014.07.14 View 15083
Artificial Antibody-based Therapeutic Candidate for Lung Cancer Developed
Professor Hak-Sung Kim of Biological Sciences at KAIST publishes a cover article on artificial antibody in "Molecular Therapy".
Repebody-based lung cancer therapeutic drug candidate developed Repebody-based protein demonstrates the possibility of the development of a new drug
KAIST Biological Sciences Department’s Professor Hak-Sung Kim, in collaboration with Professor Eun-Kyung Cho from the College of Medicine at Chungnam National University, has successfully developed an artificial antibody-based, or repebody, cancer therapeutic candidate. These research results were published as a cover paper of the July edition of Molecular Therapy.
The repebody developed by Professor Kim and his team strongly binds to interleukin-6, a cancer-causing factor. It has also been confirmed that the repebody can significantly inhibit the proliferation of cancer cells in non-small-cell lung cancer animal model.
Numerous multinational pharmaceutical and biotechnology companies have invested astronomical amounts of money in research for the development of protein therapeutics with low side effects and high efficacy. More than 20 kinds of such therapeutics are currently under clinical trials, and over 100 drugs are under clinical demonstration. Among these, the majority is antibody-based therapeutics, and most of the investments are heavily concentrated in this field. However, antibody production cost is very high because it has large molecular weights and complex structural properties, and this makes it difficult to engineer. Consequently, the development costs a great deal of time and money.
In order to overcome the existing limitations of antibody-based therapeutics, Professor Kim and his team have developed a new artificial antibody, or repebody, which was published in Proceedings of the National Academy of Sciences (PNAS) in 2012. Based on this research, they have succeeded in developing a therapeutic candidate for treating non-small-cell lung cancer with a specifically strong cohesion to the cancer-causing factor, interleukin-6.
Interleukin-6 is a crucial substance within the body that is involved in immune and inflammatory-related signals. When abnormally expressed, it activates various carcinogenic pathways and promotes tumor growth and metastasis. Because of its importance, multinational pharmaceutical companies are heavily investing in developing therapeutics that can inhibit the signaling of interleukin-6.
In this study, Professor Kim and his team observed that a repebody consists of repeated modules, and they conceived a module-based affinity amplification technology that can effectively increase the binding affinity with the disease target. The developed therapeutic candidate has been confirmed in cell and animal experiments to show low immunogenicity, as well as to strongly inhibit the proliferation of non-small-cell lung cancer.
Furthermore, by investigating the complex structure of the repebody with interleukin-6, Professor Kim has identified its mechanism, which demonstrated the potential for therapeutic development. The researchers are currently carrying out pre-clinical trials for acquiring permission to perform clinical trials on animals with non-small-cell lung cancer. The repebody can be developed into a new protein drug after demonstrating its safety and efficacy.
Professor Hak-Sung Kim and his team have confirmed that the repebody can be utilized as a new protein drug, and this will be a significant contribution to Korea’s protein drugs and biotechnology industry development.
The research was supported by the Future Pioneer Industry project and sponsored by the Ministry of Science, ICT and Future Planning.
Figure 1. Professor Kim’s article published as the cover article of July edition of Molecular Therapy
Figure 2. Clinical proof of the repebody’s inhibition of cancer growth using animal models
2014.07.14 View 15083 -
 2014 Conference on Korean Sociology Held at KAIST
The Korean Sociological Association (KSA) hosted a two-day conference in 2014 entitled “In the age of anxiety, sociology can present answers” at the College of Liberal Arts and Convergence Science, KAIST, on June 20-21, 2014. Professor Jung-Ro Yoon of the Department of Humanities and Social Sciences at KAIST is the President of KSA. The conference addressed issues such as big data and risk society. During the conference, 40 sessions took place, and 150 research papers were released.
Professor Yoon said, “The conference will offer a great opportunity for Korean sociologists to discuss anxiety, chaos, risk, and the uncertainty that Korean society experiences and suggest answers and a new vision upon which Korean society should move forward.”
2014.06.22 View 9223
2014 Conference on Korean Sociology Held at KAIST
The Korean Sociological Association (KSA) hosted a two-day conference in 2014 entitled “In the age of anxiety, sociology can present answers” at the College of Liberal Arts and Convergence Science, KAIST, on June 20-21, 2014. Professor Jung-Ro Yoon of the Department of Humanities and Social Sciences at KAIST is the President of KSA. The conference addressed issues such as big data and risk society. During the conference, 40 sessions took place, and 150 research papers were released.
Professor Yoon said, “The conference will offer a great opportunity for Korean sociologists to discuss anxiety, chaos, risk, and the uncertainty that Korean society experiences and suggest answers and a new vision upon which Korean society should move forward.”
2014.06.22 View 9223