IT
-
 KAIST Agrees to Personnel Exchange with the Ministry of Economic Affairs of the Republic of China
A delegation from the Ministry of Economic Affairs (MOEA) of the Republic of China (ROC) visited KAIST on October 16, 2015.
President Steve Kang of KAIST and Director General Yu-Ping Lien of the Department of Investment Services, MOEA, signed a memorandum of understanding (MOU) on exchanging personnel, recruiting and attracting top talents, and sharing job information.
With the MOU, KAIST and MOEA will develop a mobility program for students in Korea and ROC to help them seek employment opportunities in both nations. Director General Lien hoped that the MOU would bring more of KAIST students in the information technology field to work in ROC.
President Kang responded,
“KAIST has fostered highly talented engineers and researchers across all fields of science and engineering. If these students can have a place in ROC to realize their potential, this certainly could benefit the two nations.”
In the picture from left to right is Director General Yu-Ping Lien of the Department of Investment Services, the Ministry of Economic Affairs of the Republic of China and President Steve Kang of KAIST.
2015.10.17 View 5808
KAIST Agrees to Personnel Exchange with the Ministry of Economic Affairs of the Republic of China
A delegation from the Ministry of Economic Affairs (MOEA) of the Republic of China (ROC) visited KAIST on October 16, 2015.
President Steve Kang of KAIST and Director General Yu-Ping Lien of the Department of Investment Services, MOEA, signed a memorandum of understanding (MOU) on exchanging personnel, recruiting and attracting top talents, and sharing job information.
With the MOU, KAIST and MOEA will develop a mobility program for students in Korea and ROC to help them seek employment opportunities in both nations. Director General Lien hoped that the MOU would bring more of KAIST students in the information technology field to work in ROC.
President Kang responded,
“KAIST has fostered highly talented engineers and researchers across all fields of science and engineering. If these students can have a place in ROC to realize their potential, this certainly could benefit the two nations.”
In the picture from left to right is Director General Yu-Ping Lien of the Department of Investment Services, the Ministry of Economic Affairs of the Republic of China and President Steve Kang of KAIST.
2015.10.17 View 5808 -
 Visit by Danish Folk High School: Vallekilde Højskole
A group of 60 Danish students and teachers from Vallekilde Højskole, one of 70 Folk High Schools that are spread across Denmark, visited KAIST on October 16, 2015.
The Danish delegation and KAIST’s student organization, the International Conference for the Integration of Science, Technology, and Society (ICISTS), jointly ran a conference entitled “Learning through Having Fun and Games.”
At the conference, the Headmaster of Game Academy at Vallekilde Højskole, Thomas Vigild, gave a lecture on how Vallekilde Folk High School is educating new generations with play, curiosity, and collaboration.
During the conference, KAIST and Danish students held an event called the PlayShop, which was hosted by Headmaster Vigild. At the PlayShop, some 80 students from both institutions enjoyed Danish folk games and shared cross-cultural experiences.
The Danish Folk High School, which dates back to 1844 when its first school opened in Southern Denmark, started as a civic school offering equal learning opportunities to people less fortunate to receive a regular education.
Today, the Folk High School has grown into life-long educational institutions that provide Danish citizens from high school students to seniors with alternative, complementary education that allows citizens to venture out into new fields of their interest or further hone their professional skills and knowledge.
Established in 1865, the Vallekilde Folk High School specializes in journalism, game development, event management, literature, crafts, youth leadership, music and design. Its game development classes at the Game Academy are known for helping students reach their potential through learning from fun games, creative initiatives, and collaborative projects.
The invitation of the Danish school was made possible by Director Heekyung Park of the Institute of Disaster Studies at KAIST.
Director Park said,
“In a sense, I could say that all Danish Folk High Schools are an ideal form of delivering education. They pursue ways to teach students without losing their curiosity and interest in subjects. The schools remove any type of exams from classrooms, while striving to ensure the maximum participation of students in the learning process. KAIST could emulate some of these educational practices to offer its students a classroom free of stress and full of inspirations.”
The Danish delegation toured KAIST’s Humanoid Research Center and the Urban Robotics Lab after the conference.
2015.10.16 View 6108
Visit by Danish Folk High School: Vallekilde Højskole
A group of 60 Danish students and teachers from Vallekilde Højskole, one of 70 Folk High Schools that are spread across Denmark, visited KAIST on October 16, 2015.
The Danish delegation and KAIST’s student organization, the International Conference for the Integration of Science, Technology, and Society (ICISTS), jointly ran a conference entitled “Learning through Having Fun and Games.”
At the conference, the Headmaster of Game Academy at Vallekilde Højskole, Thomas Vigild, gave a lecture on how Vallekilde Folk High School is educating new generations with play, curiosity, and collaboration.
During the conference, KAIST and Danish students held an event called the PlayShop, which was hosted by Headmaster Vigild. At the PlayShop, some 80 students from both institutions enjoyed Danish folk games and shared cross-cultural experiences.
The Danish Folk High School, which dates back to 1844 when its first school opened in Southern Denmark, started as a civic school offering equal learning opportunities to people less fortunate to receive a regular education.
Today, the Folk High School has grown into life-long educational institutions that provide Danish citizens from high school students to seniors with alternative, complementary education that allows citizens to venture out into new fields of their interest or further hone their professional skills and knowledge.
Established in 1865, the Vallekilde Folk High School specializes in journalism, game development, event management, literature, crafts, youth leadership, music and design. Its game development classes at the Game Academy are known for helping students reach their potential through learning from fun games, creative initiatives, and collaborative projects.
The invitation of the Danish school was made possible by Director Heekyung Park of the Institute of Disaster Studies at KAIST.
Director Park said,
“In a sense, I could say that all Danish Folk High Schools are an ideal form of delivering education. They pursue ways to teach students without losing their curiosity and interest in subjects. The schools remove any type of exams from classrooms, while striving to ensure the maximum participation of students in the learning process. KAIST could emulate some of these educational practices to offer its students a classroom free of stress and full of inspirations.”
The Danish delegation toured KAIST’s Humanoid Research Center and the Urban Robotics Lab after the conference.
2015.10.16 View 6108 -
 Professor Key-Sun Choi Receives the Order of Service Merit Green Stripes from the Korean Government
The award recognizes Professor Choi’s life-long research effort to make Korean language digitally available, both nationally and internationally.
Professor Key-Sun Choi of the School of Computing at KAIST received the Order of Service Merit Green Stripes from the Korean government at the 569th Korean Language Day, held annually to commemorate the invention of the Korean language, Hangeul. The ceremony took place on October 9, 2015, at the Sejong Center in Seoul.
Professor Choi has distinguished himself in the field of natural language processing (NLP), including Korean language. He developed a Korean NLP parser that enabled information processing and data analysis of Korean language, as well as a digital Korean dictionary, contributing to the advancement of Korean language-based information technology.
Professor Choi also led the way to widespread use of Korean natural language in computing by developing and commercializing open source software to process the Korean language.
He has served leading roles in many of the international academic societies and standardization organizations, among others, as the vice president of Infoterm (the International Information Center for Terminology), president of the Asia Federation of Natural Language Processing, vice chair of ISO/TC 37, a technical committee in the International Organization for Standardization (ISO), and a council member for the International Association of Machine Translation.
2015.10.08 View 9482
Professor Key-Sun Choi Receives the Order of Service Merit Green Stripes from the Korean Government
The award recognizes Professor Choi’s life-long research effort to make Korean language digitally available, both nationally and internationally.
Professor Key-Sun Choi of the School of Computing at KAIST received the Order of Service Merit Green Stripes from the Korean government at the 569th Korean Language Day, held annually to commemorate the invention of the Korean language, Hangeul. The ceremony took place on October 9, 2015, at the Sejong Center in Seoul.
Professor Choi has distinguished himself in the field of natural language processing (NLP), including Korean language. He developed a Korean NLP parser that enabled information processing and data analysis of Korean language, as well as a digital Korean dictionary, contributing to the advancement of Korean language-based information technology.
Professor Choi also led the way to widespread use of Korean natural language in computing by developing and commercializing open source software to process the Korean language.
He has served leading roles in many of the international academic societies and standardization organizations, among others, as the vice president of Infoterm (the International Information Center for Terminology), president of the Asia Federation of Natural Language Processing, vice chair of ISO/TC 37, a technical committee in the International Organization for Standardization (ISO), and a council member for the International Association of Machine Translation.
2015.10.08 View 9482 -
 Yang-Hann Kim named recipient of the Rossing Prize in Acoustics Education by the Acoustical Society of America
Courtesy of the Acoustical Society of America (ASA)
Press release issued by ASA on October 8, 2015:
Yang-Hann Kim named recipient of the Rossing Prize in Acoustics Education by the Acoustical Society of America
Melville (NY), 8 October 2015—Yang-Hann Kim, Professor at KAIST (Korea Advanced Institute of Science and Technology), Daejeon, has been named recipient of the Acoustical Society of America (ASA) Rossing Prize in Acoustics Education. The Rossing Prize is awarded to an individual who has made significant contributions toward furthering acoustics education through distinguished teaching, creation of educational materials, textbook writing and other activities. The Prize will be presented at the 170th meeting of the ASA on 4 November 2015 in Jacksonville, Florida.
“It is my great honor to receive the Rossing Prize, which has been given to outstanding scholar members of ASA since 2003. I never dreamed to be one of them.” said Kim. “I must express my deep respect and love to my friend Thomas Rossing: I have known him more than 20 years, always respect what he has done for teaching, writing books, and pioneering work in musical acoustics.”
Yang-Hann Kim is a Fellow of the Acoustical Society of America. He received a Ph.D. from the Massachusetts Institute of Technology. His main research interests in acoustics began with “sound visualization” resulted in the development of the “sound camera” which makes any sound visible instantly. Then he moved to “sound manipulation.” Using his manipulation technology, one can move any sound in space and time, positioning sound, and can create a private sound zone. Sound Visualization and Manipulation, (Wiley, 2013), summarizes these two fields. Dr. Kim’s textbook, Sound Propagation: An Impedance Based Approach (John Wiley and Sons, 2010), is well acknowledged by the associated professional communities as one of best acoustics textbooks. Using his teaching experience at KAIST, he created a YouTube lecture on acoustics and vibration which is also available in MOOC (Massive Open Online Course). He has also presented lectures to over 500 engineers and technicians for the past 30 years.
###
The Acoustical Society of America (ASA) is the premier international scientific society in acoustics devoted to the science and technology of sound. Its 7000 members worldwide represent a broad spectrum of the study of acoustics. ASA publications include the Journal of the Acoustical Society of America—the world’s leading journal on acoustics, Acoustics Today magazine, books, and standards on acoustics. The Society also holds two major scientific meetings per year. For more information about the Society visit our website, www.acousticalsociety.org.
2015.10.06 View 10304
Yang-Hann Kim named recipient of the Rossing Prize in Acoustics Education by the Acoustical Society of America
Courtesy of the Acoustical Society of America (ASA)
Press release issued by ASA on October 8, 2015:
Yang-Hann Kim named recipient of the Rossing Prize in Acoustics Education by the Acoustical Society of America
Melville (NY), 8 October 2015—Yang-Hann Kim, Professor at KAIST (Korea Advanced Institute of Science and Technology), Daejeon, has been named recipient of the Acoustical Society of America (ASA) Rossing Prize in Acoustics Education. The Rossing Prize is awarded to an individual who has made significant contributions toward furthering acoustics education through distinguished teaching, creation of educational materials, textbook writing and other activities. The Prize will be presented at the 170th meeting of the ASA on 4 November 2015 in Jacksonville, Florida.
“It is my great honor to receive the Rossing Prize, which has been given to outstanding scholar members of ASA since 2003. I never dreamed to be one of them.” said Kim. “I must express my deep respect and love to my friend Thomas Rossing: I have known him more than 20 years, always respect what he has done for teaching, writing books, and pioneering work in musical acoustics.”
Yang-Hann Kim is a Fellow of the Acoustical Society of America. He received a Ph.D. from the Massachusetts Institute of Technology. His main research interests in acoustics began with “sound visualization” resulted in the development of the “sound camera” which makes any sound visible instantly. Then he moved to “sound manipulation.” Using his manipulation technology, one can move any sound in space and time, positioning sound, and can create a private sound zone. Sound Visualization and Manipulation, (Wiley, 2013), summarizes these two fields. Dr. Kim’s textbook, Sound Propagation: An Impedance Based Approach (John Wiley and Sons, 2010), is well acknowledged by the associated professional communities as one of best acoustics textbooks. Using his teaching experience at KAIST, he created a YouTube lecture on acoustics and vibration which is also available in MOOC (Massive Open Online Course). He has also presented lectures to over 500 engineers and technicians for the past 30 years.
###
The Acoustical Society of America (ASA) is the premier international scientific society in acoustics devoted to the science and technology of sound. Its 7000 members worldwide represent a broad spectrum of the study of acoustics. ASA publications include the Journal of the Acoustical Society of America—the world’s leading journal on acoustics, Acoustics Today magazine, books, and standards on acoustics. The Society also holds two major scientific meetings per year. For more information about the Society visit our website, www.acousticalsociety.org.
2015.10.06 View 10304 -
 Professor Kyung-Won Chung Receives the 2015 Design Value Awards
The Design Management Institute (DMI) announced eleven winners of its 2015 Design Value Awards.
Professor Kyung-Won Chung of the Industrial Design Department at KAIST took one of the four First Place Design Value Awards. The award ceremony was held on September 28, 2015, at the Royal Sonesta Hotel in Boston.
DMI is an international organization that has actively advocated for the strategic value of design, sponsoring research, publishing academic papers, connecting members across disciplines and the globe, and sharing best design practices.
Commemorating its 40th anniversary, DMI created the inaugural Design Value Awards this year, which honors the contributions of design teams who have delivered significant value through design or design management practices.
There were four categories of the awards: first, second, and third prize, and Honorable Mention. The first place had four winners; both the second and third took two winners each. The honorable mention included three awardees.
The award noted Professor Chung's contributions to the development of design management theory and practices in Korea and his dedication to elevate KAIST’s industrial design department to one of the 30 leading design institutions in the world during his 30-year career at the university.
DMI also appointed Professor Chung a Life Fellow.
Professor Chung said,
“I am greatly honored to receive such a significant award, the equivalent of the Academy Awards in the field of design. I hope this award will encourage the further development of the Korean design industry to lead the global design community.”
He has served in various important positions in public and private organizations, including as the president of the Korea Institute of Design Promotion, Vice Mayor of Design for the City of Seoul, Advisor to the Design Division of Samsung Electronics, and a member of organizing committee of the International Council of Societies of Industrial Design.
2015.10.04 View 7847
Professor Kyung-Won Chung Receives the 2015 Design Value Awards
The Design Management Institute (DMI) announced eleven winners of its 2015 Design Value Awards.
Professor Kyung-Won Chung of the Industrial Design Department at KAIST took one of the four First Place Design Value Awards. The award ceremony was held on September 28, 2015, at the Royal Sonesta Hotel in Boston.
DMI is an international organization that has actively advocated for the strategic value of design, sponsoring research, publishing academic papers, connecting members across disciplines and the globe, and sharing best design practices.
Commemorating its 40th anniversary, DMI created the inaugural Design Value Awards this year, which honors the contributions of design teams who have delivered significant value through design or design management practices.
There were four categories of the awards: first, second, and third prize, and Honorable Mention. The first place had four winners; both the second and third took two winners each. The honorable mention included three awardees.
The award noted Professor Chung's contributions to the development of design management theory and practices in Korea and his dedication to elevate KAIST’s industrial design department to one of the 30 leading design institutions in the world during his 30-year career at the university.
DMI also appointed Professor Chung a Life Fellow.
Professor Chung said,
“I am greatly honored to receive such a significant award, the equivalent of the Academy Awards in the field of design. I hope this award will encourage the further development of the Korean design industry to lead the global design community.”
He has served in various important positions in public and private organizations, including as the president of the Korea Institute of Design Promotion, Vice Mayor of Design for the City of Seoul, Advisor to the Design Division of Samsung Electronics, and a member of organizing committee of the International Council of Societies of Industrial Design.
2015.10.04 View 7847 -
 Professor Junehwa Song Appointed as the General Chair of the Organizing Committee of ACM SenSys
Professor Junehwa Song from the Schooling of Computing at KAIST has been appointed the general chair of the organizing committee of ACM SenSys—the American Computing Machine (ACM) Conference on Embedded Networked Sensor Systems.
ACM SenSys held its first conference in 2003 to promote research on wireless sensor networks and embedded systems. Since then, it has expanded into an influential international conference especially with the increasing importance in sensor technologies. Recently the committee has expanded its field of interest to mobile sensors, the Internet of Things, smart device system, and security.
Professor Song is considered a world-renown researcher in mobile and ubiquitous computing system. He presented numerous research papers at various conferences organized by ACM. He is also a member of the editorial committee of the Institute of Electrical and Electronics Engineers (IEEE) Transactions on Mobile Computing journal. For his achievements in the field and flair for coordinating and planning conferences, he is now the first Korean researcher to be appointed the chair of ACM SenSys.
Professor Song said that, as the chair, he would help discover new technology in and applications of networked, wireless sensors that would meet the demands of our modern society.
The 13th ACM SenSys will take place in Seoul—the first one to be held in Asia. The event will begin on November 1, 2015 and last four days. More information about this year’s event can be found at http://sensys.acm.org/2015/.
2015.10.02 View 8251
Professor Junehwa Song Appointed as the General Chair of the Organizing Committee of ACM SenSys
Professor Junehwa Song from the Schooling of Computing at KAIST has been appointed the general chair of the organizing committee of ACM SenSys—the American Computing Machine (ACM) Conference on Embedded Networked Sensor Systems.
ACM SenSys held its first conference in 2003 to promote research on wireless sensor networks and embedded systems. Since then, it has expanded into an influential international conference especially with the increasing importance in sensor technologies. Recently the committee has expanded its field of interest to mobile sensors, the Internet of Things, smart device system, and security.
Professor Song is considered a world-renown researcher in mobile and ubiquitous computing system. He presented numerous research papers at various conferences organized by ACM. He is also a member of the editorial committee of the Institute of Electrical and Electronics Engineers (IEEE) Transactions on Mobile Computing journal. For his achievements in the field and flair for coordinating and planning conferences, he is now the first Korean researcher to be appointed the chair of ACM SenSys.
Professor Song said that, as the chair, he would help discover new technology in and applications of networked, wireless sensors that would meet the demands of our modern society.
The 13th ACM SenSys will take place in Seoul—the first one to be held in Asia. The event will begin on November 1, 2015 and last four days. More information about this year’s event can be found at http://sensys.acm.org/2015/.
2015.10.02 View 8251 -
 KAIST and Oberthur Technologies Agree for Research and Development in Mobile Security
Professor Kwangjo Kim of the School of Computing at KAIST has signed a research and development (R&D) agreement with Marc Bertin, the Chief Technology Officer of Oberthur Technologies (OT), a French security solutions firm, on September 18, 2015 in Paris.
Under the agreement, KAIST and OT will conduct joint research on mobile security as well as implement an internship program for KAIST graduate students to work either at OT’s R&D center in Korea or in France.
OT has established a research center in Korea in July 2014, which was the fourth of its research centers in Asia.
Professor Kim said, “Our goal at KAIST is to cultivate top-level security experts in security technologies. By partnering with such a leader in security technologies as OT, we know that we will both help shape tomorrow’s security solution for the IoT (Internet of Things) space.”
In the picture, Professor Kwangjo Kim (left) shakes hands with Marc Bertin, the Chief Technology Officer of Oberthur Technologies (right), after the signing of a memorandum of understanding.
2015.09.24 View 8168
KAIST and Oberthur Technologies Agree for Research and Development in Mobile Security
Professor Kwangjo Kim of the School of Computing at KAIST has signed a research and development (R&D) agreement with Marc Bertin, the Chief Technology Officer of Oberthur Technologies (OT), a French security solutions firm, on September 18, 2015 in Paris.
Under the agreement, KAIST and OT will conduct joint research on mobile security as well as implement an internship program for KAIST graduate students to work either at OT’s R&D center in Korea or in France.
OT has established a research center in Korea in July 2014, which was the fourth of its research centers in Asia.
Professor Kim said, “Our goal at KAIST is to cultivate top-level security experts in security technologies. By partnering with such a leader in security technologies as OT, we know that we will both help shape tomorrow’s security solution for the IoT (Internet of Things) space.”
In the picture, Professor Kwangjo Kim (left) shakes hands with Marc Bertin, the Chief Technology Officer of Oberthur Technologies (right), after the signing of a memorandum of understanding.
2015.09.24 View 8168 -
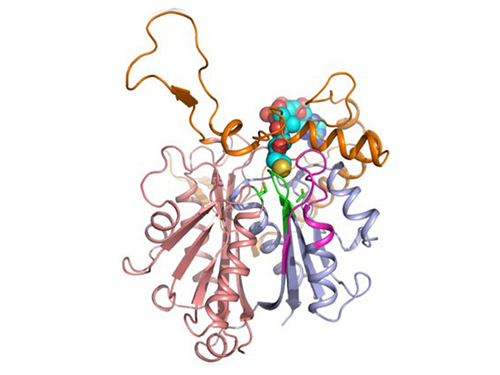 Discovery of Redox-Switch of KEenzyme Involved in N-Butanol Biosynthesis
Research teams at KAIST and Kyungpook National University (KNU) have succeeded in uncovering the redox-switch of thiolase, a key enzyme for n-butanol production in Clostridium acetobutylicum, one of the best known butanol-producing bacteria.
Biological n-butanol production was first reported by Louis Pasteur in 1861, and the bioprocess was industrialized usingClostridium acetobutylicum. The fermentation process by Clostridium strains has been known to be the most efficient one for n-butanol production. Due to growing world-wide issues such as energy security and climate change, the biological production of n-butanol has been receiving much renewed interest. This is because n-butanol possesses much better fuel characteristics compared to ethanol, such as higher energy content (29.2 MJ/L vs 19.6 MJ/L), less corrosiveness, less hygroscopy, and the ease with which it can be blended with gasoline and diesel.
In the paper published in Nature Communications, a broad-scope, online-only, and open access journal issued by the Nature Publishing Group (NPG), on September 22, 2015, Professor Kyung-Jin Kim at the School of Life Sciences, KNU, and Distinguished Professor Sang Yup Lee at the Department of Chemical and Biomolecular Engineering, KAIST, have proved that the redox-switch of thiolase plays a role in a regulation of metabolic flux in C. acetobutylicum by using in silico modeling and simulation tools.
The research team has redesigned thiolase with enhanced activity on the basis of the 3D structure of the wild-type enzyme. To reinforce a metabolic flux toward butanol production, the metabolic network of C. acetobutylicum strain was engineered with the redesigned enzyme. The combination of the discovery of 3D enzyme structure and systems metabolic engineering approaches resulted in increased n-butanol production in C. acetobutylicum, which allows the production of this important industrial chemical to be cost competitive.
Professors Kim and Lee said, "We have reported the 3D structure of C. acetobutylicum thiolase-a key enzyme involved in n-butanol biosynthesis, for the first time. Further study will be done to produce butanol more economically on the basis of the 3D structure of C. acetobutylicum thiolase."
This work was published online in Nature Communications on September 22, 2015.
Reference: Kim et al. "Redox-switch regulatory mechanism of thiolase from Clostridium acetobutylicum," Nature Communications
This research was supported by the Technology Development Program to Solve Climate Changes from the Ministry of Education, Science and Technology (MEST), Korea, the National Research Foundation of Korea, and the Advanced Biomass Center through the Global Frontier Research Program of the MEST, Korea.
For further information, contact Dr. Sang Yup Lee, Distinguished Professor, KAIST, Daejeon, Korea (leesy@kaist.ac.kr, +82-42-350-3930); and Dr. Kyung-Jin Kim, Professor, KNU, Daegu, Korea (kkim@knu.ac.kr, +82-53-950-6088).
Figure 1: A redox-switch of thiolase involves in butanol biosynthesis in Clostridium acetobutylicum. Thiolase condenses two acetyl-CoA molecules for initiating four carbon flux towards butanol.
Figure 2: Thiolase catalyzes the condensation reaction of acetyl-CoA to acetoacetyl-CoA. Two catalytic cysteine residues at 88th and 378th are oxidized and formed an intermolecular disulfide bond in an oxidized status, which results in inactivation of the enzyme for n-butanol biosynthesis. The intermolecular disulfide bond is broken enabling the n-butanol biosynthesis, when the environment status is reduced.
2015.09.23 View 11297
Discovery of Redox-Switch of KEenzyme Involved in N-Butanol Biosynthesis
Research teams at KAIST and Kyungpook National University (KNU) have succeeded in uncovering the redox-switch of thiolase, a key enzyme for n-butanol production in Clostridium acetobutylicum, one of the best known butanol-producing bacteria.
Biological n-butanol production was first reported by Louis Pasteur in 1861, and the bioprocess was industrialized usingClostridium acetobutylicum. The fermentation process by Clostridium strains has been known to be the most efficient one for n-butanol production. Due to growing world-wide issues such as energy security and climate change, the biological production of n-butanol has been receiving much renewed interest. This is because n-butanol possesses much better fuel characteristics compared to ethanol, such as higher energy content (29.2 MJ/L vs 19.6 MJ/L), less corrosiveness, less hygroscopy, and the ease with which it can be blended with gasoline and diesel.
In the paper published in Nature Communications, a broad-scope, online-only, and open access journal issued by the Nature Publishing Group (NPG), on September 22, 2015, Professor Kyung-Jin Kim at the School of Life Sciences, KNU, and Distinguished Professor Sang Yup Lee at the Department of Chemical and Biomolecular Engineering, KAIST, have proved that the redox-switch of thiolase plays a role in a regulation of metabolic flux in C. acetobutylicum by using in silico modeling and simulation tools.
The research team has redesigned thiolase with enhanced activity on the basis of the 3D structure of the wild-type enzyme. To reinforce a metabolic flux toward butanol production, the metabolic network of C. acetobutylicum strain was engineered with the redesigned enzyme. The combination of the discovery of 3D enzyme structure and systems metabolic engineering approaches resulted in increased n-butanol production in C. acetobutylicum, which allows the production of this important industrial chemical to be cost competitive.
Professors Kim and Lee said, "We have reported the 3D structure of C. acetobutylicum thiolase-a key enzyme involved in n-butanol biosynthesis, for the first time. Further study will be done to produce butanol more economically on the basis of the 3D structure of C. acetobutylicum thiolase."
This work was published online in Nature Communications on September 22, 2015.
Reference: Kim et al. "Redox-switch regulatory mechanism of thiolase from Clostridium acetobutylicum," Nature Communications
This research was supported by the Technology Development Program to Solve Climate Changes from the Ministry of Education, Science and Technology (MEST), Korea, the National Research Foundation of Korea, and the Advanced Biomass Center through the Global Frontier Research Program of the MEST, Korea.
For further information, contact Dr. Sang Yup Lee, Distinguished Professor, KAIST, Daejeon, Korea (leesy@kaist.ac.kr, +82-42-350-3930); and Dr. Kyung-Jin Kim, Professor, KNU, Daegu, Korea (kkim@knu.ac.kr, +82-53-950-6088).
Figure 1: A redox-switch of thiolase involves in butanol biosynthesis in Clostridium acetobutylicum. Thiolase condenses two acetyl-CoA molecules for initiating four carbon flux towards butanol.
Figure 2: Thiolase catalyzes the condensation reaction of acetyl-CoA to acetoacetyl-CoA. Two catalytic cysteine residues at 88th and 378th are oxidized and formed an intermolecular disulfide bond in an oxidized status, which results in inactivation of the enzyme for n-butanol biosynthesis. The intermolecular disulfide bond is broken enabling the n-butanol biosynthesis, when the environment status is reduced.
2015.09.23 View 11297 -
 KAIST and Chongqing University of Technology in China Open an International Program
With the help of KAIST, Chongqing University of Technology (CQUT) in China established an electrical engineering and computer science program and admitted their first 66 freshmen this fall semester.
The joint program was created to foster skilled engineers in the fields of electrical engineering and computer science, which are necessary for the development of the Korean and Chinese Industrial Complex located in Chongqing City.
KAIST has provided CQUT with a majority of the program’s curricula currently offered to its students in Daejeon, Korea.
Under the jointly administered program, KAIST takes on education and research while CQUT is responsible for student selection and administration.
KAIST has dispatched eight professors to teach the related fields in English, and 17 CQUT professors will teach the rest of the curricula.
In August 2014, KAIST and CQUT singed a cooperation agreement for education and research exchange and created the CQUT-KAIST Education Cooperation Center, which is headed by Professor Young-Nam Han of the Electrical Engineering Department at KAIST.
The two universities will expand their collaboration to include graduate programs by 2016.
In the picture below, President Steve Kang of KAIST (right) shakes hands with President Shi Xiaohui of Chongqing University of Technology (left).
2015.09.17 View 11419
KAIST and Chongqing University of Technology in China Open an International Program
With the help of KAIST, Chongqing University of Technology (CQUT) in China established an electrical engineering and computer science program and admitted their first 66 freshmen this fall semester.
The joint program was created to foster skilled engineers in the fields of electrical engineering and computer science, which are necessary for the development of the Korean and Chinese Industrial Complex located in Chongqing City.
KAIST has provided CQUT with a majority of the program’s curricula currently offered to its students in Daejeon, Korea.
Under the jointly administered program, KAIST takes on education and research while CQUT is responsible for student selection and administration.
KAIST has dispatched eight professors to teach the related fields in English, and 17 CQUT professors will teach the rest of the curricula.
In August 2014, KAIST and CQUT singed a cooperation agreement for education and research exchange and created the CQUT-KAIST Education Cooperation Center, which is headed by Professor Young-Nam Han of the Electrical Engineering Department at KAIST.
The two universities will expand their collaboration to include graduate programs by 2016.
In the picture below, President Steve Kang of KAIST (right) shakes hands with President Shi Xiaohui of Chongqing University of Technology (left).
2015.09.17 View 11419 -
 Professor Woontack Woo Demonstrates an Optical Platform Technology for Augmented Reality at Smart Cloud Show
Professor Woontack Woo of the Graduate School of Culture Technology at KAIST participated in the Smart Cloud Show, a technology exhibition, hosted by the university’s Augmented Human Research Center and presented the latest development of his research, an optical platform system for augmented reality.
This event took place on September 16-17, 2015 at Grand Seoul Nine Tree Convention Center in Seoul.
At the event, Professor Woo introduced a smart glass with an embedded augmented reality system, which permits remote collaboration between an avatar and the user’s hand.
The previous remote collaboration was difficult for ordinary users to employ because of its two-dimensional screen and complicated virtual reality system.
However, with the new technology, the camera attached to artificial reality (AR) glasses recognizes the user’s hand and tracks it down to collaborate. The avatar in the virtual space and the user’s hand interact in real space and time.
The key to this technology is the stable, real-time hand-tracking technique that allows the detection of the hand’s locations and the recognition of finger movements even in situations of self-occlusion.
Through this method, a user can touch and manipulate augmented contents as if they were real-life objects, thereby collaborating remotely with another user who is physically distant by linking his or her movements with an avatar.
If this technology is adopted widely, it may bring some economic benefits such as increased productivity due to lower costs for mobility and reduction in social overhead costs from the decrease in the need of traveling long distance.
Professor Woo said, “This technology will provide us with a greater opportunity for collaboration, not necessarily restricted to physical travelling, which can be widely used in the fields of medicine, education, entertainment, and tourism.”
Professor Woo plans to present his research results on hand-movement tracking and detection at the 12th International Conference on Ubiquitous Robots and Ambient Intelligence (URAI 2015), to be held on October 28-30, 2015, at Kintex in Goyang, Korea.
He will also present a research paper on remote collaboration at the ICAT-EGVE 2015 conference, the merger of the 25th International Conference on Artificial Reality and Telexistence (ICAT 2015) and the 20th Eurographics Symposium on Virtual Environments (EGVE 2015), which will take place on October 28-30, 2015 at the Kyoto International Community House, Kyoto, Japan.
2015.09.16 View 9813
Professor Woontack Woo Demonstrates an Optical Platform Technology for Augmented Reality at Smart Cloud Show
Professor Woontack Woo of the Graduate School of Culture Technology at KAIST participated in the Smart Cloud Show, a technology exhibition, hosted by the university’s Augmented Human Research Center and presented the latest development of his research, an optical platform system for augmented reality.
This event took place on September 16-17, 2015 at Grand Seoul Nine Tree Convention Center in Seoul.
At the event, Professor Woo introduced a smart glass with an embedded augmented reality system, which permits remote collaboration between an avatar and the user’s hand.
The previous remote collaboration was difficult for ordinary users to employ because of its two-dimensional screen and complicated virtual reality system.
However, with the new technology, the camera attached to artificial reality (AR) glasses recognizes the user’s hand and tracks it down to collaborate. The avatar in the virtual space and the user’s hand interact in real space and time.
The key to this technology is the stable, real-time hand-tracking technique that allows the detection of the hand’s locations and the recognition of finger movements even in situations of self-occlusion.
Through this method, a user can touch and manipulate augmented contents as if they were real-life objects, thereby collaborating remotely with another user who is physically distant by linking his or her movements with an avatar.
If this technology is adopted widely, it may bring some economic benefits such as increased productivity due to lower costs for mobility and reduction in social overhead costs from the decrease in the need of traveling long distance.
Professor Woo said, “This technology will provide us with a greater opportunity for collaboration, not necessarily restricted to physical travelling, which can be widely used in the fields of medicine, education, entertainment, and tourism.”
Professor Woo plans to present his research results on hand-movement tracking and detection at the 12th International Conference on Ubiquitous Robots and Ambient Intelligence (URAI 2015), to be held on October 28-30, 2015, at Kintex in Goyang, Korea.
He will also present a research paper on remote collaboration at the ICAT-EGVE 2015 conference, the merger of the 25th International Conference on Artificial Reality and Telexistence (ICAT 2015) and the 20th Eurographics Symposium on Virtual Environments (EGVE 2015), which will take place on October 28-30, 2015 at the Kyoto International Community House, Kyoto, Japan.
2015.09.16 View 9813 -
 Professor Seok-Jung Kang Is Appointed the Director of the Korea Institute of Ceramic Engineering and Technology
Professor Seok-Jung Kang of the Material Sciences and Engineering Department at KAIST has been appointed to the position of the third Director of the Korea Institute of Ceramic Engineering and Technology. His three-year term will begin September 9, 2015.
An authority in the field of ceramics, Professor Kang has taught at KAIST since 1980. He received his bachelor’s degree from the Department of Metallurgical Engineering at Seoul National University, his master’s degree at KAIST, and his doctoral degree at the Ecole Centrale de Paris.
Professor Kang received the Robert B. Sosman Award in 2011, an annual prize given by the American Society of Ceramics to recognize outstanding achievement in basic science in an area that results in significant impact to the field of ceramics. He was the first Asian recipient of the award.
He also received the 2015 Professor Sir Richard Brooke Award, a prestigious bi-annual prize presented to an outstanding scholar by the European Society of Ceramics.
2015.09.15 View 7078
Professor Seok-Jung Kang Is Appointed the Director of the Korea Institute of Ceramic Engineering and Technology
Professor Seok-Jung Kang of the Material Sciences and Engineering Department at KAIST has been appointed to the position of the third Director of the Korea Institute of Ceramic Engineering and Technology. His three-year term will begin September 9, 2015.
An authority in the field of ceramics, Professor Kang has taught at KAIST since 1980. He received his bachelor’s degree from the Department of Metallurgical Engineering at Seoul National University, his master’s degree at KAIST, and his doctoral degree at the Ecole Centrale de Paris.
Professor Kang received the Robert B. Sosman Award in 2011, an annual prize given by the American Society of Ceramics to recognize outstanding achievement in basic science in an area that results in significant impact to the field of ceramics. He was the first Asian recipient of the award.
He also received the 2015 Professor Sir Richard Brooke Award, a prestigious bi-annual prize presented to an outstanding scholar by the European Society of Ceramics.
2015.09.15 View 7078 -
 KAIST Participates in the World Economic Forum's Annual Meeting of the New Champions 2015 in China
KAIST’s president and its professors actively engage in discussions of major issues on higher education, technology innovation, and industry-university collaboration with global leaders from across all sectors.
President Steve Kang of KAIST participated in the Annual Meeting of the New Champions 2015 (a.k.a., Summer Davos Forum) hosted by the World Economic Forum (WEF). With the theme of “Charting a New Course for Growth,” the Summer Davos Forum took place on September 9-11, 2015 in, Dalian, China.
Currently, KAIST is a member of the Global University Leaders Forum (GULF) of WEF, a gathering of the presidents of the top 25 universities in the world, including Harvard University, Massachusetts Institute of Technology, University of Tokyo, University of Oxford, Peking University, and National University of Singapore. GULF allows university leaders an opportunity to have high-level dialogues on higher education and research and explore prospects for cooperative ventures.
President Kang led the discussion of the GULF session at the Summer Davos Forum, which was held on September 10, 2015, with 25 university leaders as well as two business leaders from Chinese companies: Huawei Technologies Co. Ltd., and Sanofi China. The participants shared candid perspectives on industry-university collaboration, particularly the need for such partnerships in Asia.
In addition, KAIST hosted the fourth IdeasLab session, entitled “Bio versus Nano Materials, on September 9, 2015. At the session, four KAIST professors held an in-depth debate and discussion with the audience on whether the next industrial revolution would be driven by advances in biomaterials or nanomaterials.
The topics under discussion were:
- New materials that mimic biology by Professor Hea Shin Lee
- Bio-based materials that replace petroleum-based materials by Professor Sang Yup Lee
- New materials designed at sub-nano scale by Professor Hee Tae Jung
- A hydrogen economy with nanomaterials by Professor Eun Ae Cho
Since its establishment in 2007, the Summer Davos Forum has become the biggest business and political gathering in Asia, held annually either in Dalian or Tianjin, China. The Forum has attracted more than 1,500 participants primarily from emerging nations such as China, India, Russia, Mexico, and Brazil, and has offered an open platform to address issues important to the region and the global community.
2015.09.14 View 9138
KAIST Participates in the World Economic Forum's Annual Meeting of the New Champions 2015 in China
KAIST’s president and its professors actively engage in discussions of major issues on higher education, technology innovation, and industry-university collaboration with global leaders from across all sectors.
President Steve Kang of KAIST participated in the Annual Meeting of the New Champions 2015 (a.k.a., Summer Davos Forum) hosted by the World Economic Forum (WEF). With the theme of “Charting a New Course for Growth,” the Summer Davos Forum took place on September 9-11, 2015 in, Dalian, China.
Currently, KAIST is a member of the Global University Leaders Forum (GULF) of WEF, a gathering of the presidents of the top 25 universities in the world, including Harvard University, Massachusetts Institute of Technology, University of Tokyo, University of Oxford, Peking University, and National University of Singapore. GULF allows university leaders an opportunity to have high-level dialogues on higher education and research and explore prospects for cooperative ventures.
President Kang led the discussion of the GULF session at the Summer Davos Forum, which was held on September 10, 2015, with 25 university leaders as well as two business leaders from Chinese companies: Huawei Technologies Co. Ltd., and Sanofi China. The participants shared candid perspectives on industry-university collaboration, particularly the need for such partnerships in Asia.
In addition, KAIST hosted the fourth IdeasLab session, entitled “Bio versus Nano Materials, on September 9, 2015. At the session, four KAIST professors held an in-depth debate and discussion with the audience on whether the next industrial revolution would be driven by advances in biomaterials or nanomaterials.
The topics under discussion were:
- New materials that mimic biology by Professor Hea Shin Lee
- Bio-based materials that replace petroleum-based materials by Professor Sang Yup Lee
- New materials designed at sub-nano scale by Professor Hee Tae Jung
- A hydrogen economy with nanomaterials by Professor Eun Ae Cho
Since its establishment in 2007, the Summer Davos Forum has become the biggest business and political gathering in Asia, held annually either in Dalian or Tianjin, China. The Forum has attracted more than 1,500 participants primarily from emerging nations such as China, India, Russia, Mexico, and Brazil, and has offered an open platform to address issues important to the region and the global community.
2015.09.14 View 9138